According to Facebook announcement by Svitlana Panaiotidi, state authorized person of the AMCU, on February 21st, 2019 the AMCU accepted advisory explanations on application on economic competition protection legislation by participants of medicines market in vertical relations for supply and promotion of medicines.
Advisory explanation is a summarized practice of the AMCU in cases considered by the competition authority regarding pharmaceutical companies, distributors and pharmacies.
The announcement states that the Advisory Explanation consists of the following sections:
1. Legal analysis;
2. Peculiarities of pharmaceutical market functioning;
3. Market limits determination;
4. Risks of violating legislation on protection of economic competition:
Svitlana Panaiotidi in her announcement stated that the effects described in the Advisory Explanations can relate also to other types of violations of economic competition protection legislation.
On February 26th, 2019 (while this comment was being prepared) full text of the Advisory Explanations appeared on the AMCU website.
Therefore, instead of the short comment, we offer you full text of the Advisory Explanations.
Section “Legal Regulation” of the Advisory Explanations contains key norms used by the AMCU to assess vertical concerted actions of the pharmaceutical market participants. In particular, these are article 6 of the Law of Ukraine “On Protection of Economic Competition” which defines anticompetitive concerted actions, and article 8 which contains exceptions from article 6 related to agreements on supply or use of goods.
Further, the AMCU refers to the key provisions of the Standard Requirements to vertical concerted actions of commercial entities related to supply and use of goods approved by Decree of the AMCU No. 10-рп as of October 12th, 2017. It, in particular, reminds that vertical concerted actions are allowed if share of each participant (both supplier and buyer) at the corresponding market of contractual goods does not exceed 30%, as well as draws attention to some strict vertical restrictions risky for competition irrespective of the concerted actions participants’ share amounts.
In Section II devoted to peculiarities of the pharmaceutical market functioning, the AMCU described market participants and some peculiarities of each market. In particular, the AMCU has specified that manufacturers, importers and distributors engage in wholesale trade in medicines at the national market, and retail medicine markets are regional ones.
Characterizing the distribution market, the AMCU noted that during the recent years two biggest domestic distributors have had significant market power at the market of wholesale medicines and medical devices batches supply of wide range in distribution/whole sale trade sectors and have not faced significant competition at this market due to relatively small amount of shares owned by competitors.
Describing medicines retail market, the AMCU noted that these markets are highly competitive at the national level, however can be described as monopoly (dominant position)/collective monopoly of some commercial entities at regional levels.
One of the most interesting parts of the Advisory Explanations is Section III devoted to determination of commodity limits of markets of medicines. Svitlana Panaiotidi made announcement of the approaches described in this section in the article published in Yurydychna Gazeta of February 25th, 2019.
Hence, pursuant to the Advisory Explanations and based on the conditions of realization and prescription of medicines, the AMCU will be guided by the following approaches to determining the commodity limits of medicines markets:
1. Interchangeable medicines will compose a single commodity market due to the fact that they have the same active substance, dosage and dose form (generic approach) in the following cases:
A.When it concerns markets of medicines realization through public procurement procedures;
B.When it concerns markets of prescription medicines realization, where influence of end consumer on market is not decisive.
2. In other cases interchangeable medicines will compose a single commodities market based on wider understanding of medicines interchangeability (for example, their therapeutic interchangeability (АТХ‑3)). In particular, it is possible for OTC medicines outside public procurement system purported for removal/relieving symptoms of some simple diseases, and also when a consumer chooses a medicine (without doctor participation) and for own money. There a qualified consumer demand and bona fide seller (pharmacist, etc.) supply are taken into consideration.
The abovementioned criteria specified by the AMCU to application of various approaches to determine commodity limits of medicines markets are, in our opinion, quite reasonable.
Such approach of the AMCU to determining commodity limits of markets is more liberal compared to the approach applied by this authority within the completed investigations, and to the maximum extent close to the approaches use by our company in its practice.
Further, it is recommended to consider the Advisory Explanations as general landmarks in the AMCU approaches to asses some matters. In particular, we believe that the Advisory Explanations allow to apply a wide approach to determining commodity limits of the market to a number of prescription medicines chosen to be purchased by the end consumer. For example, it can be valid for prescription medicines improving quality of life. The brightest example can be prescription medicines against erectile dysfunction. Unfortunately, we do not have statistical data of doctors prescribing such medicines, however we can assume that in most cases a patient decides both to use and buy such prescription medicines.
It should be noted that the described approach currently does not cover competition between manufacturers in the sector called “competition for medical professionals’ brains”. This sector is extremely complex and multifaceted and in different cases can have own peculiarities, therefore we can hardly expect any general approaches from the AMCU to consider it during determination of commodity limits of medicines markets.
Section IV of the Advisory Explanations contains description of the most discussed concerted actions taking place on the pharmaceutical market and which have repeatedly been included in the AMCU decisions, namely:
Discounts. The most important aspect to pay attention to is a list of anticompetitive discount effects, namely:
The AMCU also specifies in what cases motivating sales in relations with distributors can be seen as violation of legislation on economic competition protection:
The AMCU with respect to stimulation of sales volumes states that when market share of supplier and/or buyer is equal to or exceeds 30%, vertical concerted actions providing for stimulating the buyer by the supplier to increase good realization volume by granting discounts to the buyer aimed at encouraging buyers distributors to increase medicines realization volumes can be allowed under article 10 of the Law on condition that it does not lead to removal and/or non-acceptance of other manufacturers’ goods from/to buyers distributors’ portfolio.
The increased risk of violating legislation on economic competition protection is mixing in one list for discount granting of unique medicines and medicines having substitutes.
The violation can be use of discounts during procurement of some medicines provided in case of procurement of other medicines. In particular, if on markets of medicines for which discounts are granted the supplier faces competition, and on markets of medicines to purchase of which the discount is due the supplier takes monopoly (dominant) position, discount granting entails restriction of competition on markets where it is faced by the supplier.
While application of discounts to prevent violations of legislation on economic competition protection, participants of medicines markets are advised by the AMCU to consider the following:
Risk assessment matrix contained in the Advisory Explanation is quite illustrative:
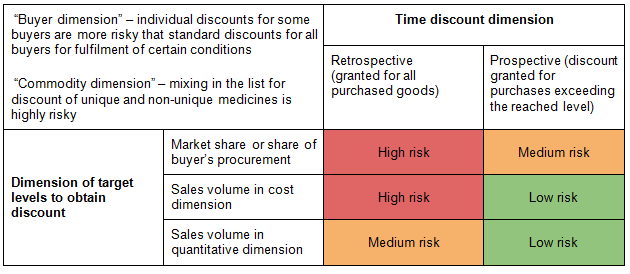
The following provisions of vertical agreements increase risk of anticompetitive effect possibility during discount setting:
The following influences determination of anticompetitive effect possibility level:
Reporting. While setting conditions for distributor reporting to suppliers, in order to prevent violations of economic competition protection legislation, the AMCU recommends being guided by the following:
Patient support programs. During implementation of the patient support programs the AMCU recommends keeping the following principles:
Besides it should be considered that the patient support programs:
It should be noted that the pharmaceutical market needs this sort of summary and explanation of the AMCU approaches to competition risks in relations among manufacturers of medicines (their representatives), distributors and pharmacies, since the competition legislation contains quite general requirements, which complicates their application without comprehensive specialized examination.
The Advisory Explanations have outlined safe areas and determined danger signals to let market participants analyse competitive risks alone in simple cases, and professionals have been granted a basis to rely on in case of profound analysis of this risk category.
Please address your antitrust & competition questions to: Andriy Gorbatenko, partner, gorbatenko@l-a.com.ua
The event focused on the transformation of Ukraine's intellectual property sector on its path toward European integration.
Improving access to safe and affordable medicines for the Ukrainian population is one of the Government of Ukraine's top priorities. SAFEMed Activity (2017-2025) has supported this effort by appIying health system strengthening best practices.
The Ministry of Health website has posted a notice about the release of a revised draft order of the Ministry of Health ‘On Approval of Amendments to Certain Re
On 25 September, a webinar was held on the topic: ‘180 days of new drug price regulation. Results, prospects and practical advice.’ The event was organized by LA Law Firm in partnership with Proxima Research International.